Biosimilars, generics among tools that can help ensure employee health
Health care remains a fundamental and defining issue for Canadians. As such, ensuring that private drug plans evolve with Canada’s changing health-care landscape is of great importance.
It is critical that plan sponsors have the right information to ensure employee health benefits continue to support the best possible health outcomes for the workforce, while ensuring sustainability as the rising costs of medications remains a constant challenge.
We are not just living longer in Canada, we’re now working longer, too. Chronic diseases and increased utilization are contributing factors to growing prescription claims costs amid the already high costs of drug plans in Canada.
As we continue to see the unique impacts of our country’s aging population, it is important for plan sponsors to prepare for the need to provide adequate drug coverage for aging employees and to ensure plans are designed to support drug plan sustainability.
The TELUS Health Drug Data Trends and National Benchmarks Report highlights trends in Canadian private drug plan costs impacted by regulatory issues, particularly those affecting employee benefits, as well as providing insights on utilization and plan design tool adoption.
It uncovers the story behind prescription drug claims, the multiple industries and communities it affects and how we can better use and analyze the data to help build sustainable benefits plans.
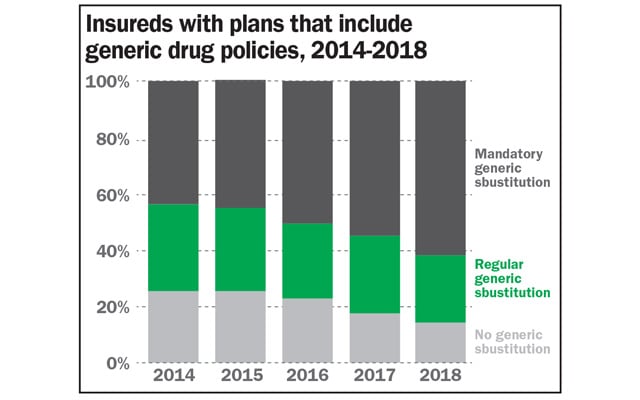
Challenges with mandatory policies
Further efforts to improve plan sustainability should look at plan design tools related to the use of generic drugs and biosimilars. Biosimilars are alternatives to first-on-market specialty biologic drugs made from or containing living organisms, which often exhibit complex molecular structures.
Despite there being no expected clinically meaningful differences in efficacy and safety between a biosimilar and the biologic drug that was already authorized for sale, as well as them being substantially less expensive, uptake has not been consistent across drug types and therapeutic areas.
Hesitancy around the use of biosimilars is proving to be a major barrier to the adoption of mandatory switch policies that would provide the same benefits to patients while reducing eligible costs.
Mandatory switch policies have the potential to significantly increase the acceleration of biosimilars uptake, creating savings that could potentially be invested into other drugs and treatment areas, contributing to overall plan sustainability.
Public policymakers have recognized the benefits of biosimilars adoption, with British Columbia leading the charge as the first public payer to implement a mandatory switch policy in early 2019.
While mandatory switch policies may be viewed as too aggressive or disruptive for some plan sponsors, additional options exist through the use of prior authorization or step therapy approaches where newly diagnosed members must start with the biosimilar.
Another option for plan sponsors is a reference-based pricing type approach, where reimbursement for the originator is limited to that of the biosimilar. This is an option for preserving plan member choice while, at the same time, providing cost containment for the plan.
Generics and plan sustainability
Policies for generic drugs can also play a role in improving plan sustainability. Generics are bioequivalent to their originators in terms of chemical composition and clinical benefits, but they are available at more affordable prices.
As private plan sponsors aim to design long-lasting plans, they may consider options for favouring the increased use of generic molecules over their multisource brands or me-too, single-source molecules.
This can be done through a number of ways, including plan design changes, formulary management and generic substitution options. Approaches could include current mandatory generic options as well as broader generic substitutions that go beyond the traditional approaches of interchangeability. This can allow plan sponsors to capitalize on some of the interchangeability nuances in provinces such as British Columbia and Alberta.
In light of Canada’s advancing health-care landscape, plan sponsors should take a closer look at their own drug plan data, collaborate with trusted advisers and develop insights-driven health benefits plans that continue to support the workforce of today and tomorrow.
The use of biosimilars and generics are two of the tools in an ever-evolving landscape that can help ensure employee health while building sustainable drug plans of the future.
Together, with a commitment to thoughtful assessment of both patient outcomes and cost, we can imagine and achieve this careful but critical balance.
Jason Kennedy is a pharmacist and director of operations at TELUS Health. For more information, visit www.telus.com/en/health.




